
Using molar masses and unit conversions, we are able to find the grams of solute (NaCl) and solvent (Water). To start this problem, we need to convert our information into the same unit, in this case, grams. Let’s walk through an example below to further understand this calculation! Percent by Weight Exampleĭetermine the percent by weight of sodium chloride in the following solution: 1.75 moles of NaCl (58.44 g/mol) dissolved in. To calculate the mass percent composition (or simply, the mass percent) of an element in a compound, we divide the mass of the element in 1 mol of the compound. Lastly, you plug the numbers into the percent by weight equation and get your answer. You should already have the grams of solute, so you can use the same methods to find the grams of solvent. Determine the quality of the mixture in Only the final answer must be in 3 decimal points. The mass of the liquid is found to be 15 kg. 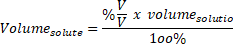
A newly purchased container that has a capacity of 1 m³ contains a mixture of liquid and steam in equilibrium at a temperature of 601 K. Then, you have to find the total grams of solution, which includes the solvent and solute. Determine the percent vapor on a mass basis (x). From now on, we will refer to the units in grams to stay consistent. You may also be given an amount in moles, which you can convert by using the molar mass. For example, if an amount is given in milligrams and the other in kilograms, you will have to convert using the correct conversions. Using Molar Mass calculations to figure the percentage that. Sometimes the amounts in grams will be given, but sometimes you might have to do some extra work to get it to the same units. Objective 4: Calculate the percentage composition of a given chemical compound. However, each method ends the same way: you need to find the ratio of how many grams of solute there are for the total grams of solution.įirst, when doing any calculation, you have to make sure you are using consistent units. If you have any queries about percent composition formula, drop a comment below and we will get back to you.When doing this calculation, the way you arrive at your answer depends on the information given to you. Question: How does increasing the concentration of sugar in water effect the mass of potato cubes Hypothesis: If. We hope this article on Mass Percent Formula has helped you. Its value is represented by the percentage. Definitions: Theoretical Yield: The maximum amount of product if ALL of the limiting reagent reacted exactly as described by the balanced equation. What is the unit of percent by mass?Īns: Percent by mass as no unit. Mass of solution \( = \) Mass of solute \( \) Mass of solvent percent v/v solution is calculated by the following formula using the milliliter as the base measure of volume (v): v/v mL of solute/100 mL of solution. In this measurement system, the concentration of a solution is expressed as the mass of solute in grams present per \(100\, \times 100\) Here, crystals of common salt undergo a physical state change from solid to fine particles attached to water molecules. The solution composition can be described in mass percentage which shows the mass of solute present in a given mass of solution. (ii) Solvent: The substance that dissolves the other substance and does not undergo a change in its physical state is called a solvent.įor example, when common salt is dissolved in water, common salt is a solute, and water is a solvent. (i) Solute: The substance that is being dissolved or undergoes a change in its physical state is called a solute. A solution essentially contains two components: Volume by volume percentage of solute in solution.Ī solution is a homogeneous mixture of two or more substances.Mass by volume percentage of solute in solution.Mass by the mass percentage of solute in solution.The concentration of a solution is expressed in several ways such as: The concentration of a solution is defined as the amount of solute present per unit mass or per unit volume of the solution. This can apply to any sort of chemical mixture but most often is used in relation to solutions. The comparison is done in the form of masses, not by the volume. Although mass percent is unit-less it can be given with units as well in special cases. 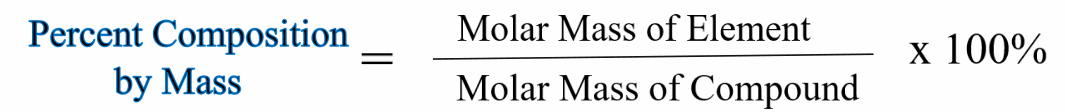
The mass percentage is just an excellent style for presenting the concentration of an element within a compound or component of the mixture. In chemistry, concentration is the measure of how much of a given substance is mixed with another substance. The mass percent formula in Chemistry is given as.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
